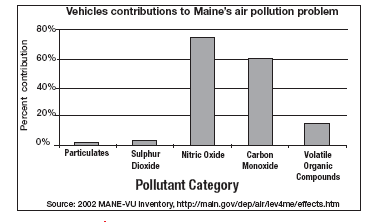
Use the above image to answer question 1
_____ 1. Based on your knowledge about acid rain, which two air pollutants from vehicles would contribute most to the production of acid precipitation in the state of Maine?
a. sulfur dioxide and nitric oxide
b. nitric oxide and carbon monoxide
c. carbon monoxide and volatile organic compounds
d. sulfur dioxide and volatile organic compounds
_____ 2. When as acid is added to pure water, it results in
a. an increase in the water’s pH
b. a greater concentration of OH- ions
c. a greater concentration of H+ ions
d. a neutralization reaction
_____ 3. Acid precipitation with a pH of 4 is
a. 3 times more acid than pure water
b. 10 times more acid than pure water
c. 100 times more acid than pure water
d. 1000 times more acid than pure water
_____ 1. Based on your knowledge about acid rain, which two air pollutants from vehicles would contribute most to the production of acid precipitation in the state of Maine?
a. sulfur dioxide and nitric oxide
b. nitric oxide and carbon monoxide
c. carbon monoxide and volatile organic compounds
d. sulfur dioxide and volatile organic compounds
_____ 2. When as acid is added to pure water, it results in
a. an increase in the water’s pH
b. a greater concentration of OH- ions
c. a greater concentration of H+ ions
d. a neutralization reaction
_____ 3. Acid precipitation with a pH of 4 is
a. 3 times more acid than pure water
b. 10 times more acid than pure water
c. 100 times more acid than pure water
d. 1000 times more acid than pure water
 RSS Feed
RSS Feed
