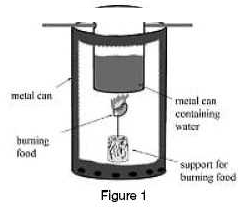
1. What is the correct sequence of energy conversions occurring as the calorimeter is being used?
a. thermal energy --> chemical energy --> mechanical energy
b. mechanical energy --> chemical energy --> thermal energy
c. chemical energy --> thermal energy --> mechanical energy
d. mechanical energy --> thermal energy --> chemical energy
2. The way the Calorimeter works is similar to the way most power plants function. However, in a power plant
a. the burning fuel is not used to heat water
b. no heat is lost to the surrounding environment
c. chemical energy is converted directly to electricity
d. steam turns turbines, which drive electric generators
3. One type of power plant that does not involve the combustion of a fuel is a
a. petroleum powered facility
b. nuclear powered facility
c. biomass powered facility
d. natural gas powered facility
a. thermal energy --> chemical energy --> mechanical energy
b. mechanical energy --> chemical energy --> thermal energy
c. chemical energy --> thermal energy --> mechanical energy
d. mechanical energy --> thermal energy --> chemical energy
2. The way the Calorimeter works is similar to the way most power plants function. However, in a power plant
a. the burning fuel is not used to heat water
b. no heat is lost to the surrounding environment
c. chemical energy is converted directly to electricity
d. steam turns turbines, which drive electric generators
3. One type of power plant that does not involve the combustion of a fuel is a
a. petroleum powered facility
b. nuclear powered facility
c. biomass powered facility
d. natural gas powered facility
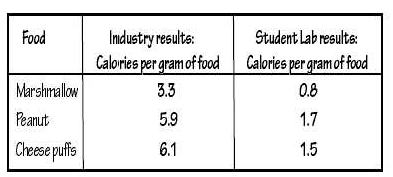
When the students who built the Calorimeter in Figure 1 compared their calculations to the industrial Caloric values printed on food labels they noticed some differences. Study the data in the table below and answer questions 1 and 2.
1. Explain two possible reasons for the differences between the lab values and the industrial values.
2. How could the students change the Calorimeter’s design to improve the accuracy of the results?
1. Explain two possible reasons for the differences between the lab values and the industrial values.
2. How could the students change the Calorimeter’s design to improve the accuracy of the results?
 RSS Feed
RSS Feed
